Yara Merhi - Google Review
Dr. Timani is very thorough and highly informed. She didn't rush or interrupt me while I was talking. I loved the results of my treatment. Highly recommend this practice.
PATIENT REVIEWS


Indications, Important Safety Information and Prescribing Information
Indications, Important Safety Information and Prescribing
Information
BOTOX® Cosmetic
(onabotulinumtoxinA) Important Information
Indications
BOTOX® Cosmetic (onabotulinumtoxinA) is
indicated in adult patients for the temporary improvement in the appearance of:
- Moderate to severe glabellar lines associated with
corrugator and/or procerus muscle activity
- Moderate to severe lateral canthal lines associated with
orbicularis oculi activity
- Moderate to severe forehead lines associated with frontalis
activity
IMPORTANT SAFETY INFORMATION, INCLUDING BOXED WARNING
WARNING: DISTANT SPREAD OF TOXIN EFFECT
Postmarketing reports indicate that the effects of BOTOX® Cosmetic
and all botulinum toxin products may spread from the area of injection to
produce symptoms consistent with botulinum toxin effects. These may include
asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia,
dysarthria, urinary incontinence, and breathing difficulties. These symptoms
have been reported hours to weeks after injection. Swallowing and breathing
difficulties can be life threatening and there have been reports of death. The
risk of symptoms is probably greatest in children treated for spasticity, but
symptoms can also occur in adults treated for spasticity and other conditions,
particularly in those patients who have an underlying condition that would
predispose them to these symptoms. In unapproved uses and approved indications,
cases of spread of effect have been reported at doses comparable to those used
to treat cervical dystonia and spasticity and at lower doses.
CONTRAINDICATIONS
BOTOX® Cosmetic is contraindicated in the
presence of infection at the proposed injection site(s) and in individuals with
known hypersensitivity to any botulinum toxin preparation or to any of the
components in the formulation.
WARNINGS AND PRECAUTIONS
Lack of Interchangeability Between Botulinum Toxin
Products
The potency units of BOTOX® Cosmetic are
specific to the preparation and assay method utilized. They are not interchangeable
with other preparations of botulinum toxin products and, therefore, units of
biological activity of BOTOX® Cosmetic cannot be compared to
nor converted into units of any other botulinum toxin products assessed with
any other specific assay method.
Spread of Toxin Effect
Please refer to Boxed Warning for Distant Spread of Toxin
Effect.
No definitive serious adverse event reports of distant
spread of toxin effect associated with dermatologic use of BOTOX® Cosmetic
at the labeled dose of 20 Units (for glabellar lines), 24 Units (for lateral
canthal lines), 40 Units (for forehead lines with glabellar lines), 44 Units
(for simultaneous treatment of lateral canthal lines and glabellar lines), and
64 Units (for simultaneous treatment of lateral canthal lines, glabellar lines,
and forehead lines) have been reported. Patients or caregivers should be
advised to seek immediate medical care if swallowing, speech, or respiratory
disorders occur.
Serious Adverse Reactions With Unapproved Use
Serious adverse reactions, including excessive weakness,
dysphagia, and aspiration pneumonia, with some adverse reactions associated
with fatal outcomes, have been reported in patients who received BOTOX® injections
for unapproved uses. In these cases, the adverse reactions were not necessarily
related to distant spread of toxin, but may have resulted from the
administration of BOTOX® to the site of injection and/or
adjacent structures. In several of the cases, patients had pre-existing
dysphagia or other significant disabilities. There is insufficient information
to identify factors associated with an increased risk for adverse reactions
associated with the unapproved uses of BOTOX®. The safety and
effectiveness of BOTOX® for unapproved uses have not been
established.
Hypersensitivity Reactions
Serious and/or immediate hypersensitivity reactions have
been reported. These reactions include anaphylaxis, serum sickness, urticaria,
soft-tissue edema, and dyspnea. If such reactions occur, further injection of
BOTOX® Cosmetic should be discontinued and appropriate medical
therapy immediately instituted. One fatal case of anaphylaxis has been reported
in which lidocaine was used as the diluent and, consequently, the causal agent
cannot be reliably determined.
Cardiovascular System
There have been reports following administration of BOTOX® of
adverse events involving the cardiovascular system, including arrhythmia and
myocardial infarction, some with fatal outcomes. Some of these patients had
risk factors including pre-existing cardiovascular disease. Use caution when
administering to patients with pre-existing cardiovascular disease.
Increased Risk of Clinically Significant Effects With
Pre-existing Neuromuscular Disorders
Individuals with peripheral motor neuropathic diseases,
amyotrophic lateral sclerosis, or neuromuscular junction disorders (eg,
myasthenia gravis or Lambert-Eaton syndrome) should be monitored when given
botulinum toxin. Patients with neuromuscular disorders may be at increased risk
of clinically significant effects including generalized muscle weakness,
diplopia, ptosis, dysphonia, dysarthria, severe dysphagia, and respiratory
compromise from onabotulinumtoxinA (see Warnings and
Precautions).
Dysphagia and Breathing Difficulties
Treatment with BOTOX® and other botulinum
toxin products can result in swallowing or breathing difficulties. Patients
with pre-existing swallowing or breathing difficulties may be more susceptible
to these complications. In most cases, this is a consequence of weakening of
muscles in the area of injection that are involved in breathing or
oropharyngeal muscles that control swallowing or breathing (see Boxed
Warning).
Pre-existing Conditions at the Injection Site
Caution should be used when BOTOX® Cosmetic
treatment is used in the presence of inflammation at the proposed injection
site(s) or when excessive weakness or atrophy is present in the target
muscle(s).
Dry Eye in Patients Treated With BOTOX® Cosmetic
There have been reports of dry eye associated with BOTOX® Cosmetic
injection in or near the orbicularis oculi muscle. If symptoms of dry eye (eg,
eye irritation, photophobia, or visual changes) persist, consider referring
patients to an ophthalmologist.
Human Albumin and Transmission of Viral Diseases
This product contains albumin, a derivative of human blood.
Based on effective donor screening and product manufacturing processes, it
carries an extremely remote risk for transmission of viral diseases and variant
Creutzfeldt-Jakob disease (vCJD). There is a theoretical risk for transmission
of Creutzfeldt-Jakob disease (CJD), but if that risk actually exists, the risk
of transmission would also be considered extremely remote. No cases of
transmission of viral diseases, CJD or vCJD have ever been identified for
licensed albumin or albumin contained in other licensed products.
ADVERSE REACTIONS
The most frequently reported adverse reactions following
injection of BOTOX® Cosmetic for glabellar lines were eyelid
ptosis (3%), facial pain (1%), facial paresis (1%), and muscular weakness (1%).
The most frequently reported adverse reaction following
injection of BOTOX® Cosmetic for lateral canthal lines was
eyelid edema (1%).
The most frequently reported adverse reactions following
injection of BOTOX® Cosmetic for forehead lines with glabellar
lines were headache (9%), brow ptosis (2%), and eyelid ptosis (2%).
DRUG INTERACTIONS
Co-administration of BOTOX® Cosmetic and
aminoglycosides or other agents interfering with neuromuscular transmission
(eg, curare-like compounds) should only be performed with caution as the effect
of the toxin may be potentiated. Use of anticholinergic drugs after
administration of BOTOX® Cosmetic may potentiate systemic
anticholinergic effects.
The effect of administering different botulinum neurotoxin
products at the same time or within several months of each other is unknown.
Excessive neuromuscular weakness may be exacerbated by
administration of another botulinum toxin prior to the
resolution of the effects of a previously administered botulinum toxin.
Excessive weakness may also be exaggerated by administration
of a muscle relaxant before or after administration of BOTOX® Cosmetic.
USE IN SPECIFIC POPULATIONS
There are no studies or adequate data from postmarketing
surveillance on the developmental risk associated with use of BOTOX® Cosmetic
in pregnant women. There are no data on the presence of BOTOX® Cosmetic
in human or animal milk, the effects on the breastfed child, or the effects on
milk production.
Please see BOTOX® Cosmetic
full Prescribing
Information including Boxed Warning and Medication Guide.
Looking for Botox in Sandy Springs? At Springs Dermatology MD, Botox®, Dysport®, and other FDA-approved wrinkle relaxers are performed by Dr. Shereen Timani, a triple board-certified dermatologist. Our goal is simple: soften lines while preserving natural expression — so you look refreshed, never frozen.
We accept the Allē Loyalty Program for Botox.
Why Choose a Dermatologist for Botox?
When it comes to injectables, precision matters. Botox is a medical procedure that requires deep knowledge of facial anatomy, muscle dynamics, and dosing balance. At Springs Dermatology MD:
• Treatments are performed by a triple board-certified dermatologist
• Unit-based dosing ensures precise, customized results
• Strict sterile protocols are followed
• We prioritize subtle, balanced enhancement
• Every treatment plan evolves with your facial structure and goals
Unlike high-volume medspas, your care here is physician-led from consultation to injection.
What Does Botox Treat?
Botox works best on dynamic wrinkles, lines created by repeated facial movement.
Common treatment areas include:
• Frown lines between the brows
• Forehead lines
• Crow’s feet Bunny lines
• Chin dimpling
• Neck bands
Advanced Uses of Botox
• Lip flip to subtly enhance the upper lip
• Jawline slimming and facial contouring
• Neck band relaxation (Nefertiti lift)
• Micro-dosing for pore refinement
• Treatment support for facial flushing
• Each treatment is customized based on anatomy and goals.
While Botox is primarily used for dynamic wrinkles, it may also play a role in select cases of facial flushing. Learn more about how we approach rosacea with personalized, dermatologist-led care.
How Long Does Botox Last?
Most patients begin noticing changes within 48 hours, with full results visible around 10–14 days.
Results typically last 3–4 months, depending on muscle strength and metabolism. With consistent treatment, many patients notice longer-lasting smoothing over time.
How Many Units of Botox Will I Need?
There is no “one-size-fits-all” number.
Units depend on:
• Muscle strength
• Treatment area
• Desired degree of movement
• Facial symmetry
You are charged only for the exact number of units used, no flat pricing or bundled guesswork.
Botox vs. Dermal Fillers: What’s the Difference?
Botox relaxes the muscles that create dynamic expression lines, such as frown lines and crow’s feet. Dermal fillers, on the other hand, restore lost volume, enhance contours, and soften deeper folds that are present even at rest. While Botox treats movement-related wrinkles, fillers address volume loss.
During your consultation, we determine which approach — or combination — best fits your goals, often as part of a broader cosmetic dermatology treatment plan.
Will Botox Look Natural?
Yes, when performed correctly. Dr. Timani's philosophy is refinement, not exaggeration. Botox should soften expression lines while allowing you to look like yourself. During consultation, we discuss your goals carefully and adjust dosing accordingly.
Is Botox Safe?
Botox has been used for decades and is FDA-approved for cosmetic wrinkle reduction.
When administered by a qualified physician in a controlled medical setting, it has a strong safety profile. As with any medical treatment, risks and benefits are discussed thoroughly during consultation.
Additional Wrinkle Relaxer Options
In addition to Botox, we offer:
Dysport®
Xeomin®
Each product has subtle differences in diffusion and onset time. We guide you toward the best option for your anatomy and goals.
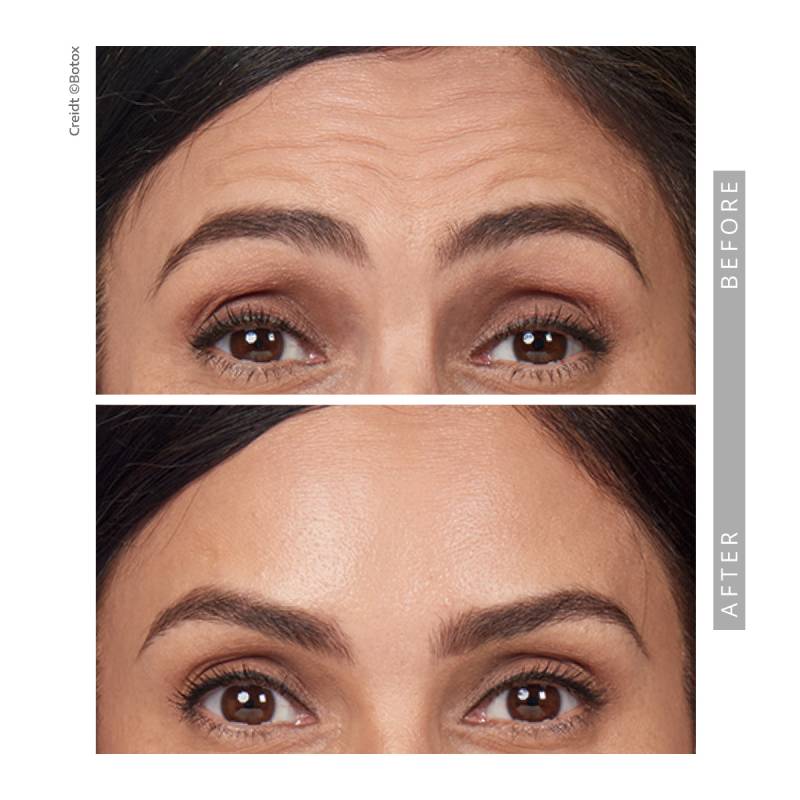
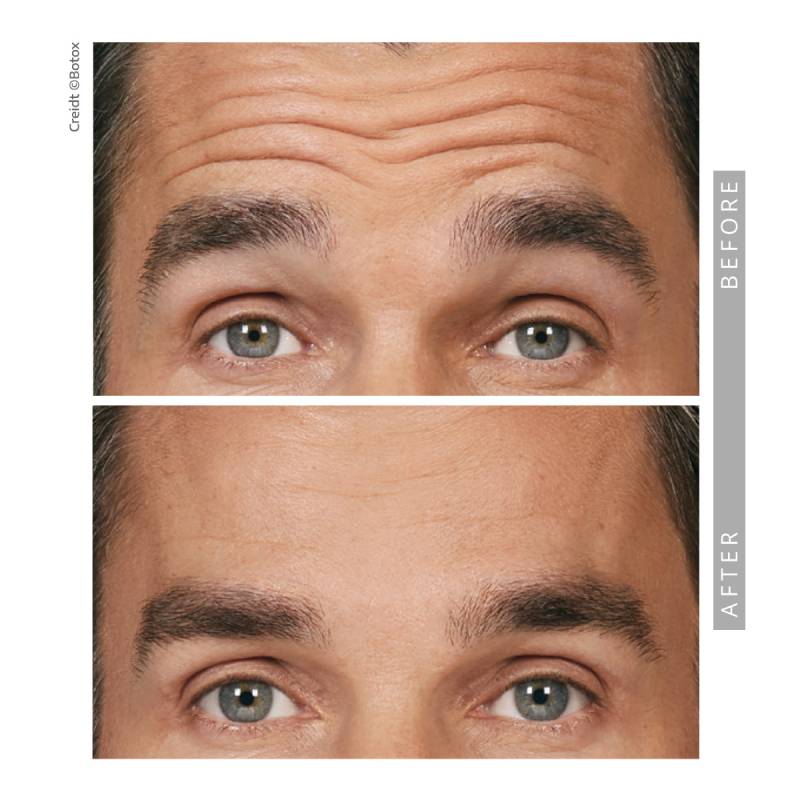
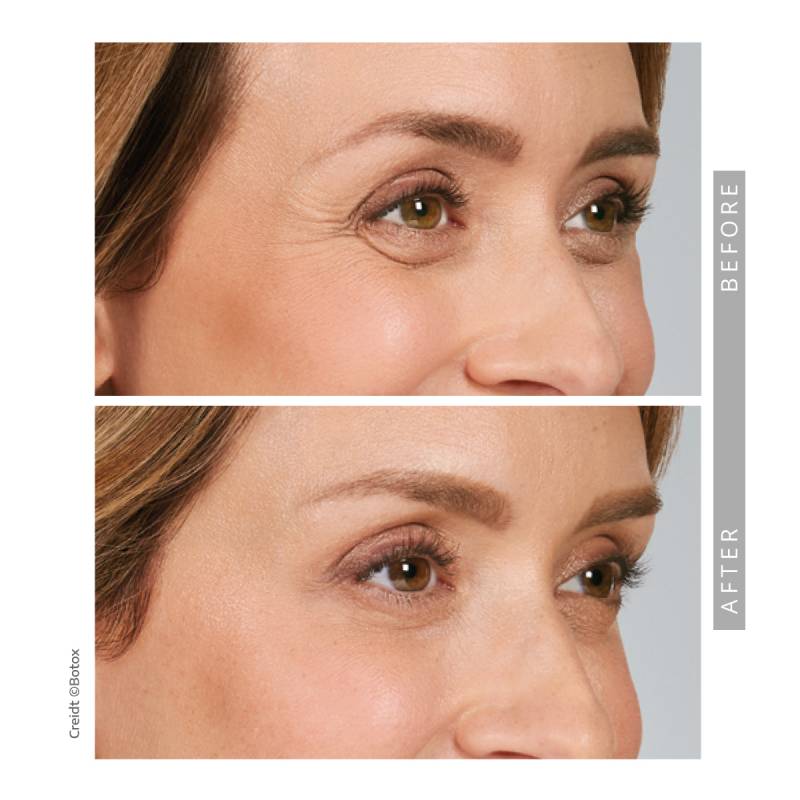
Before & After Results
We invite you to explore our before-and-after gallery to see real patient results. Subtle, balanced, and natural outcomes define our approach.
Botox for Men
Men increasingly seek Botox to maintain a competitive and refreshed appearance. Treatment plans are customized to preserve masculine features while softening lines.
What to Expect During Your Appointment
• Consultation and facial assessment
• Customized dosing plan
• Quick injection session
• Minimal downtime
Most patients return to normal activity the same day.
Serving Sandy Springs & Metro Atlanta
Our Sandy Springs office serves patients from Atlanta, Dunwoody, Roswell, Brookhaven, Marietta, Alpharetta, and surrounding communities.
Convenient access and physician-led care set us apart.
Frequently Asked Questions
Does Botox hurt?
Most patients tolerate injections very well. We use techniques that minimize discomfort.
When will I see results?
Initial changes may appear within 48 hours. Full results develop in about 2 weeks.
Can I prevent wrinkles with Botox?
Some patients choose preventative treatment to soften early expression lines before they deepen.
Is there downtime?
There is minimal to no downtime. Specific aftercare instructions are provided.
Schedule Your Botox Consultation
If you’re considering Botox in Sandy Springs, we invite you to schedule a consultation with Dr. Timani to discuss a customized treatment plan designed around your facial structure and goals.
Call (470) 769-9400 to book your appointment.
Indications, Important Safety Information and Prescribing Information
Indications, Important Safety Information and Prescribing
Information
BOTOX® Cosmetic
(onabotulinumtoxinA) Important Information
Indications
BOTOX® Cosmetic (onabotulinumtoxinA) is
indicated in adult patients for the temporary improvement in the appearance of:
- Moderate to severe glabellar lines associated with
corrugator and/or procerus muscle activity
- Moderate to severe lateral canthal lines associated with
orbicularis oculi activity
- Moderate to severe forehead lines associated with frontalis
activity
IMPORTANT SAFETY INFORMATION, INCLUDING BOXED WARNING
WARNING: DISTANT SPREAD OF TOXIN EFFECT
Postmarketing reports indicate that the effects of BOTOX® Cosmetic
and all botulinum toxin products may spread from the area of injection to
produce symptoms consistent with botulinum toxin effects. These may include
asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia,
dysarthria, urinary incontinence, and breathing difficulties. These symptoms
have been reported hours to weeks after injection. Swallowing and breathing
difficulties can be life threatening and there have been reports of death. The
risk of symptoms is probably greatest in children treated for spasticity, but
symptoms can also occur in adults treated for spasticity and other conditions,
particularly in those patients who have an underlying condition that would
predispose them to these symptoms. In unapproved uses and approved indications,
cases of spread of effect have been reported at doses comparable to those used
to treat cervical dystonia and spasticity and at lower doses.
CONTRAINDICATIONS
BOTOX® Cosmetic is contraindicated in the
presence of infection at the proposed injection site(s) and in individuals with
known hypersensitivity to any botulinum toxin preparation or to any of the
components in the formulation.
WARNINGS AND PRECAUTIONS
Lack of Interchangeability Between Botulinum Toxin
Products
The potency units of BOTOX® Cosmetic are
specific to the preparation and assay method utilized. They are not interchangeable
with other preparations of botulinum toxin products and, therefore, units of
biological activity of BOTOX® Cosmetic cannot be compared to
nor converted into units of any other botulinum toxin products assessed with
any other specific assay method.
Spread of Toxin Effect
Please refer to Boxed Warning for Distant Spread of Toxin
Effect.
No definitive serious adverse event reports of distant
spread of toxin effect associated with dermatologic use of BOTOX® Cosmetic
at the labeled dose of 20 Units (for glabellar lines), 24 Units (for lateral
canthal lines), 40 Units (for forehead lines with glabellar lines), 44 Units
(for simultaneous treatment of lateral canthal lines and glabellar lines), and
64 Units (for simultaneous treatment of lateral canthal lines, glabellar lines,
and forehead lines) have been reported. Patients or caregivers should be
advised to seek immediate medical care if swallowing, speech, or respiratory
disorders occur.
Serious Adverse Reactions With Unapproved Use
Serious adverse reactions, including excessive weakness,
dysphagia, and aspiration pneumonia, with some adverse reactions associated
with fatal outcomes, have been reported in patients who received BOTOX® injections
for unapproved uses. In these cases, the adverse reactions were not necessarily
related to distant spread of toxin, but may have resulted from the
administration of BOTOX® to the site of injection and/or
adjacent structures. In several of the cases, patients had pre-existing
dysphagia or other significant disabilities. There is insufficient information
to identify factors associated with an increased risk for adverse reactions
associated with the unapproved uses of BOTOX®. The safety and
effectiveness of BOTOX® for unapproved uses have not been
established.
Hypersensitivity Reactions
Serious and/or immediate hypersensitivity reactions have
been reported. These reactions include anaphylaxis, serum sickness, urticaria,
soft-tissue edema, and dyspnea. If such reactions occur, further injection of
BOTOX® Cosmetic should be discontinued and appropriate medical
therapy immediately instituted. One fatal case of anaphylaxis has been reported
in which lidocaine was used as the diluent and, consequently, the causal agent
cannot be reliably determined.
Cardiovascular System
There have been reports following administration of BOTOX® of
adverse events involving the cardiovascular system, including arrhythmia and
myocardial infarction, some with fatal outcomes. Some of these patients had
risk factors including pre-existing cardiovascular disease. Use caution when
administering to patients with pre-existing cardiovascular disease.
Increased Risk of Clinically Significant Effects With
Pre-existing Neuromuscular Disorders
Individuals with peripheral motor neuropathic diseases,
amyotrophic lateral sclerosis, or neuromuscular junction disorders (eg,
myasthenia gravis or Lambert-Eaton syndrome) should be monitored when given
botulinum toxin. Patients with neuromuscular disorders may be at increased risk
of clinically significant effects including generalized muscle weakness,
diplopia, ptosis, dysphonia, dysarthria, severe dysphagia, and respiratory
compromise from onabotulinumtoxinA (see Warnings and
Precautions).
Dysphagia and Breathing Difficulties
Treatment with BOTOX® and other botulinum
toxin products can result in swallowing or breathing difficulties. Patients
with pre-existing swallowing or breathing difficulties may be more susceptible
to these complications. In most cases, this is a consequence of weakening of
muscles in the area of injection that are involved in breathing or
oropharyngeal muscles that control swallowing or breathing (see Boxed
Warning).
Pre-existing Conditions at the Injection Site
Caution should be used when BOTOX® Cosmetic
treatment is used in the presence of inflammation at the proposed injection
site(s) or when excessive weakness or atrophy is present in the target
muscle(s).
Dry Eye in Patients Treated With BOTOX® Cosmetic
There have been reports of dry eye associated with BOTOX® Cosmetic
injection in or near the orbicularis oculi muscle. If symptoms of dry eye (eg,
eye irritation, photophobia, or visual changes) persist, consider referring
patients to an ophthalmologist.
Human Albumin and Transmission of Viral Diseases
This product contains albumin, a derivative of human blood.
Based on effective donor screening and product manufacturing processes, it
carries an extremely remote risk for transmission of viral diseases and variant
Creutzfeldt-Jakob disease (vCJD). There is a theoretical risk for transmission
of Creutzfeldt-Jakob disease (CJD), but if that risk actually exists, the risk
of transmission would also be considered extremely remote. No cases of
transmission of viral diseases, CJD or vCJD have ever been identified for
licensed albumin or albumin contained in other licensed products.
ADVERSE REACTIONS
The most frequently reported adverse reactions following
injection of BOTOX® Cosmetic for glabellar lines were eyelid
ptosis (3%), facial pain (1%), facial paresis (1%), and muscular weakness (1%).
The most frequently reported adverse reaction following
injection of BOTOX® Cosmetic for lateral canthal lines was
eyelid edema (1%).
The most frequently reported adverse reactions following
injection of BOTOX® Cosmetic for forehead lines with glabellar
lines were headache (9%), brow ptosis (2%), and eyelid ptosis (2%).
DRUG INTERACTIONS
Co-administration of BOTOX® Cosmetic and
aminoglycosides or other agents interfering with neuromuscular transmission
(eg, curare-like compounds) should only be performed with caution as the effect
of the toxin may be potentiated. Use of anticholinergic drugs after
administration of BOTOX® Cosmetic may potentiate systemic
anticholinergic effects.
The effect of administering different botulinum neurotoxin
products at the same time or within several months of each other is unknown.
Excessive neuromuscular weakness may be exacerbated by
administration of another botulinum toxin prior to the
resolution of the effects of a previously administered botulinum toxin.
Excessive weakness may also be exaggerated by administration
of a muscle relaxant before or after administration of BOTOX® Cosmetic.
USE IN SPECIFIC POPULATIONS
There are no studies or adequate data from postmarketing
surveillance on the developmental risk associated with use of BOTOX® Cosmetic
in pregnant women. There are no data on the presence of BOTOX® Cosmetic
in human or animal milk, the effects on the breastfed child, or the effects on
milk production.
Please see BOTOX® Cosmetic
full Prescribing
Information including Boxed Warning and Medication Guide.
Yara Merhi - Google Review
Dr. Timani is very thorough and highly informed. She didn't rush or interrupt me while I was talking. I loved the results of my treatment. Highly recommend this practice.
PATIENT REVIEWS