Peggy Wood - Google Review
Dr Timani is an artist. Providing state of the art treatments that are natural looking. Never overdone. She is a perfectionist sand takes her time to make each treatment perfect.
PATIENT REVIEWS
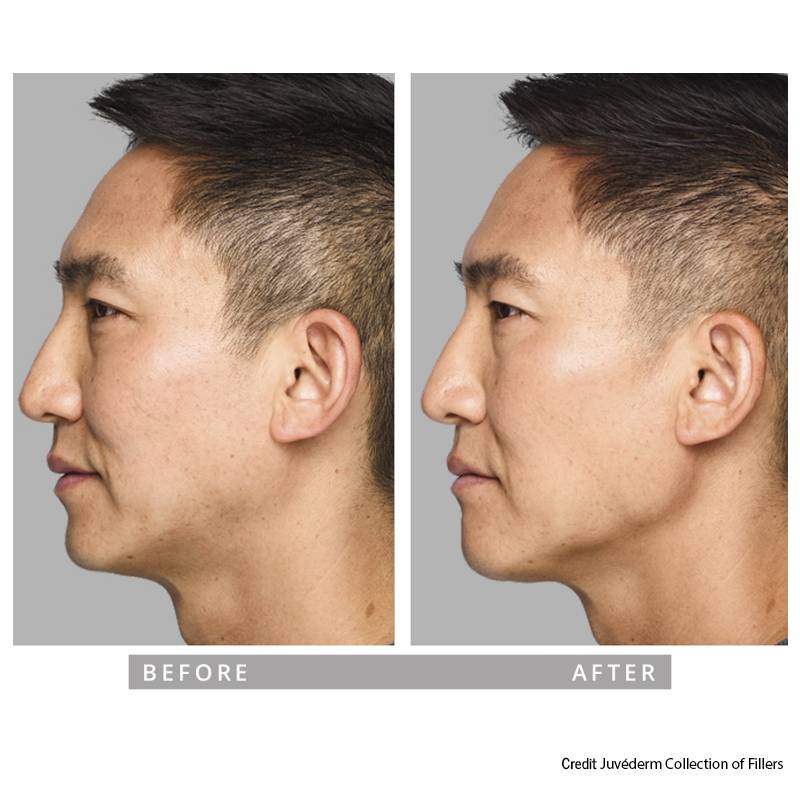
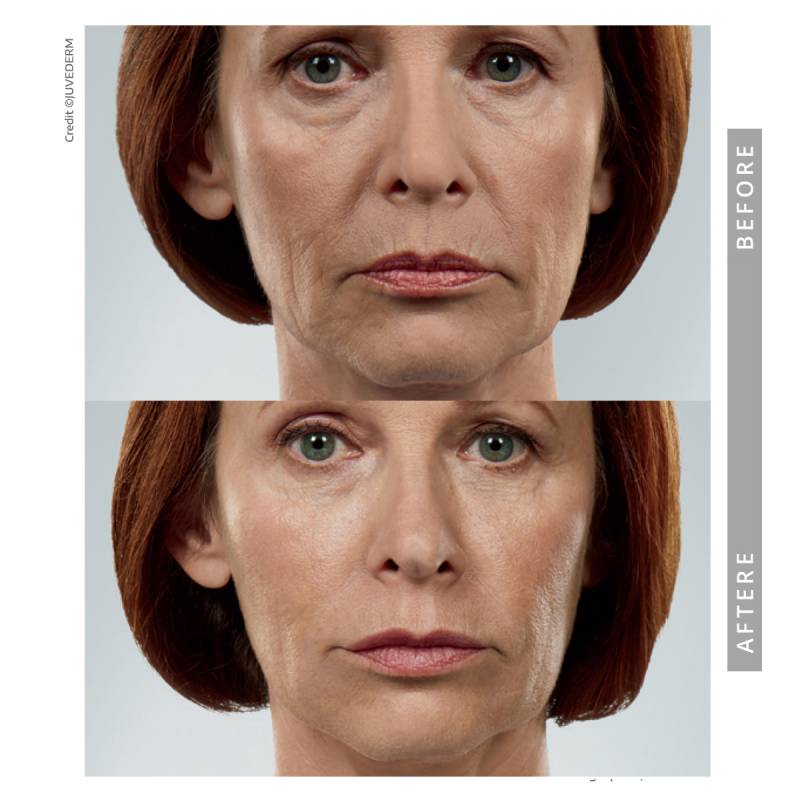
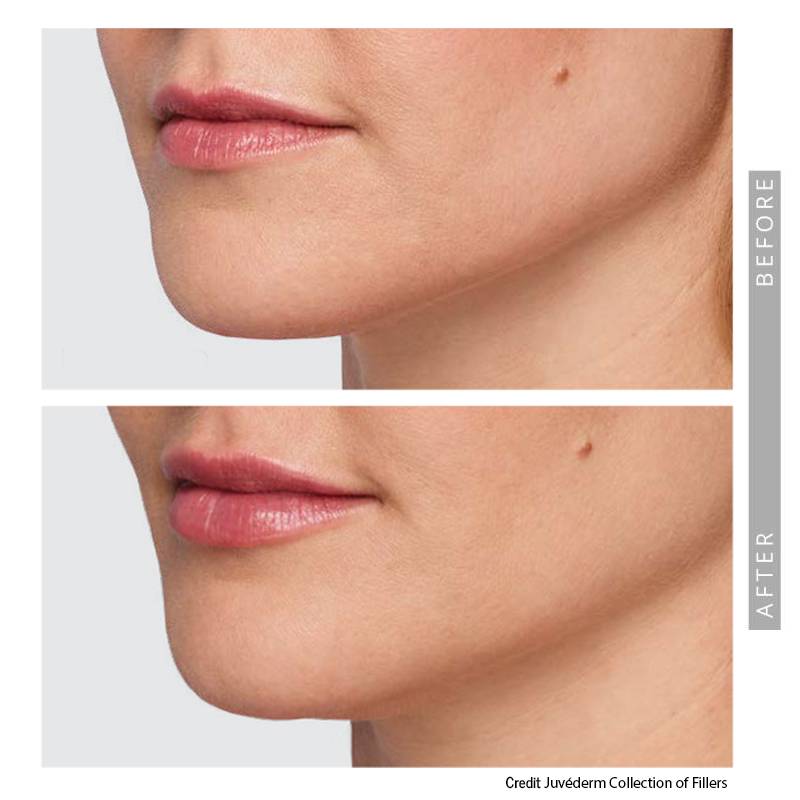
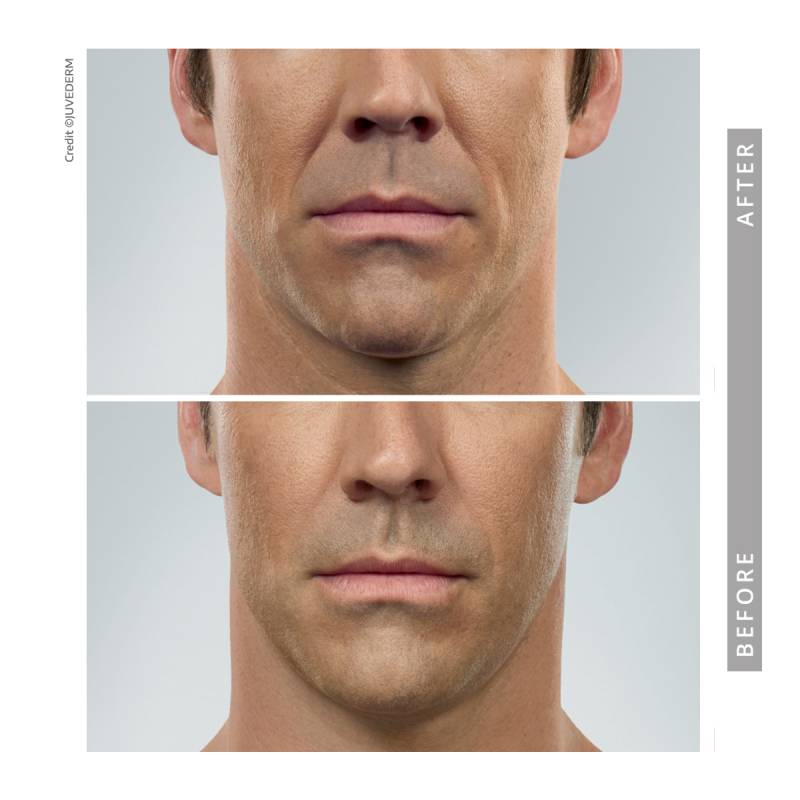

Indications, Important Safety Information and Prescribing Information
All images © 2020 Allergan
JUVÉDERM® Collection of Fillers Important Information
INDICATIONS
JUVÉDERM® VOLUMA™ XC injectable gel is indicated for deep (subcutaneous and/or supraperiosteal) injection for cheek augmentation to correct age-related volume deficit in the mid-face in adults over the age of 21.
JUVÉDERM® VOLLURE™ XC injectable gel is indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds) in adults over the age of 21.
JUVÉDERM® Ultra Plus XC and JUVÉDERM® Ultra XC injectable gels are indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds).
JUVÉDERM® VOLBELLA™ XC injectable gel is indicated for injection into the lips for lip augmentation and for correction of perioral rhytids in adults over the age of 21.
JUVÉDERM® Ultra XC injectable gel is indicated for injection into the lips and perioral area for lip augmentation in adults over the age of 21.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
These products should not be used in patients who have severe allergies, marked by a history of anaphylaxis or history or presence of multiple severe allergies, and should not be used in patients with a history of allergies to Gram-positive bacterial proteins or lidocaine contained in these products.
WARNINGS
- Do not inject into blood vessels. Introduction of these products into the vasculature may lead to embolization, occlusion of the vessels, ischemia, or infarction. Take extra care when injecting soft-tissue fillers; for example, inject the product slowly and apply the least amount of pressure necessary. Rare, but serious, adverse events associated with the intravascular injection of soft-tissue fillers in the face have been reported and include temporary or permanent vision impairment, blindness, cerebral ischemia or cerebral hemorrhage leading to stroke, skin necrosis, and damage to underlying facial structures. Immediately stop the injection if a patient exhibits any of the following symptoms: changes in vision, signs of a stroke, blanching of the skin, unusual pain during or shortly after the procedure. Patients should receive prompt medical attention and, possibly, evaluation by an appropriate healthcare professional specialist should an intravascular injection occur
- Product use at specific sites in which an active inflammatory process (skin eruptions such as cysts, pimples, rashes, or hives) or infection is present should be deferred until the underlying process has been controlled
PRECAUTIONS
- To minimize the risk of potential complications, these products should only be used by healthcare professionals who have appropriate training, experience, and knowledge of facial anatomy
- Healthcare professionals are encouraged to discuss the potential risks of soft-tissue injections with their patients prior to treatment and ensure that patients are aware of signs and symptoms of potential complications
- The safety and effectiveness for the treatment of anatomic regions other than the mid-face with JUVÉDERM® VOLUMA™ XC; facial wrinkles and folds with JUVÉDERM® Ultra XC, JUVÉDERM® Ultra Plus XC, and JUVÉDERM® VOLLURE™ XC; and the lips and perioral area with JUVÉDERM® Ultra XC and JUVÉDERM® VOLBELLA™ XC have not been established in controlled clinical studies
- As with all transcutaneous procedures, dermal filler implantation carries a risk of infection. Follow standard precautions associated with injectable materials
- The safety for use during pregnancy, in breastfeeding females, and in patients with known susceptibility to keloid formation, hypertrophic scarring, and pigmentation disorders has not been studied
- The safety for use of JUVÉDERM® VOLUMA™ XC in patients under 35 or over 65 years, JUVÉDERM® Ultra XC and JUVÉDERM® Ultra Plus XC in patients under 18 years, and JUVÉDERM® VOLLURE™ XC and JUVÉDERM® VOLBELLA™ XC in patients under 22 years has not been established
- Use with caution in patients on immunosuppressive therapy
- Patients who are using products that can prolong bleeding (such as aspirin, nonsteroidal anti-inflammatory drugs, and warfarin) may experience increased bruising or bleeding at treatment sites
- If laser treatment, chemical peel, or any other procedure based on active dermal response is considered after treatment, or if these products are administered before the skin has healed completely, there is a possible risk of an inflammatory reaction at the treatment site
- Patients who experience skin injury near the site of implantation may be at a higher risk for adverse events
- The safety of JUVÉDERM® VOLUMA™ XC injectable gel for use in patients with very thin skin in the mid-face and Fitzpatrick Skin Types V and VI has not been established
- Patients may experience late onset nodules with use of dermal fillers including JUVÉDERM® VOLUMA™ XC
- Patients may experience late onset adverse events with use of dermal fillers
ADVERSE EVENTS
The most commonly reported side effects for JUVÉDERM® injectable gels were injection-site redness, swelling, pain, tenderness, firmness, lumps/bumps, bruising, discoloration, and itching. For JUVÉDERM® VOLBELLA™ XC, dryness was also reported. For JUVÉDERM® VOLUMA™ XC, side effects were predominantly moderate in severity, with duration of 2 to 4 weeks; for JUVÉDERM® Ultra XC , JUVÉDERM® Ultra Plus XC, or JUVÉDERM® VOLLURE™ XC, they were mostly mild or moderate in severity, with duration of 14 days or less; and for JUVÉDERM® VOLBELLA™ XC, they were predominantly mild or moderate, with duration of 30 days or less.
To report an adverse reaction with any product in the JUVÉDERM® Collection, please call Allergan at 1-800-433-8871. Please visit JuvedermDFU.com for more information.
Products in the JUVÉDERM® Collection are available by prescription only.
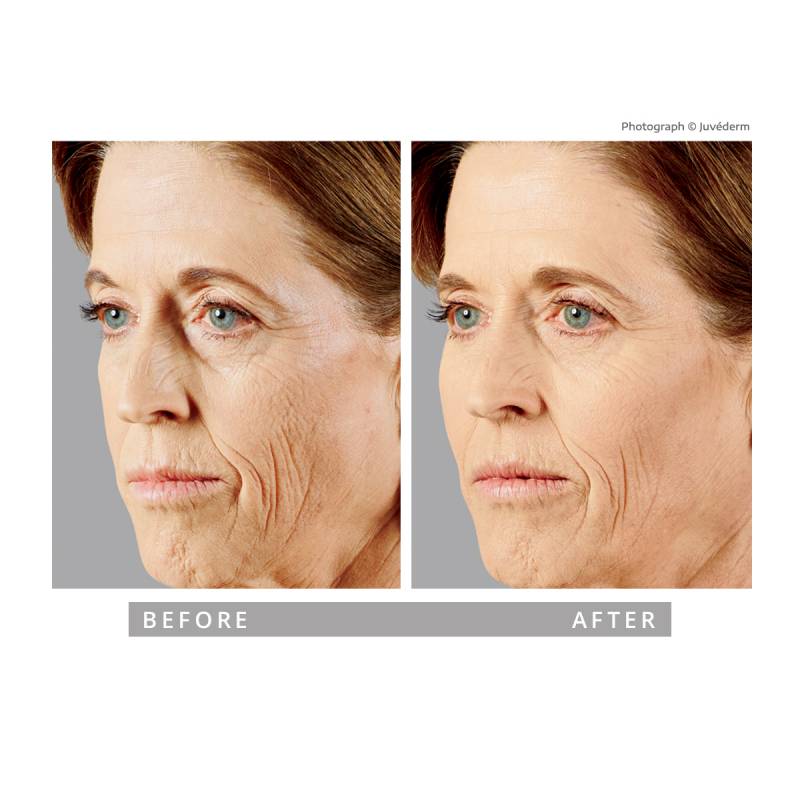
Refined, natural dermal and lip filler results that still look like you
At Springs Dermatology MD in Sandy Springs, dermal filler treatments are performed exclusively by Dr. Shereen Timani, a triple board-certified dermatologist. Every injection is guided by advanced knowledge of facial anatomy, dermatology training, and a conservative aesthetic philosophy.
We focus on restoring volume, enhancing facial balance, and maintaining natural expression — never overfilled, never artificial.
Filler Options, Selected for Precision and Balance
We use a curated range of FDA-approved dermal fillers and collagen-supporting injectables, selected based on your anatomy, goals, and the area being treated. Techniques may include cannula-based placement when appropriate.
Juvéderm® Family
(including Skinvive™, Voluma®, Vollure®, Volux®, Volbella®)
A collection of hyaluronic acid fillers designed for lips, cheeks, jawline definition, and skin hydration. Different formulations vary in flexibility and structure depending on the treatment area.
Restylane®
Hyaluronic acid fillers known for precision and versatility, often selected for subtle contouring and natural movement.
Sculptra®
An injectable option that supports gradual collagen restoration over time, commonly used for deeper structural support.
Radiesse®
A calcium-based filler that provides immediate volume and may also support collagen over time in select areas.
We proudly accept the Allē Loyalty Program for Botox®, Juvéderm®, and Skinvive™ treatments.
Why Patients in Sandy Springs Choose Dr. Timani for Our Fillers
Dermal fillers require far more than product knowledge — they demand medical expertise, artistic judgment, and precision.
Artistry and Precision
All treatments are performed by Dr. Shereen Timani, a triple board-certified dermatologist, known for meticulous technique and conservative, anatomy-based aesthetic care.
Personalized Consultations
Every treatment begins with a detailed consultation. We tailor each plan to your facial structure, goals, and comfort level—never a one-size-fits-all approach.
Proven, Patient-Centered Results
Patients trust us for subtle refinement or more noticeable rejuvenation—always with realistic expectations and a focus on long-term facial harmony.
Safety and Transparency
We use only FDA-approved dermal fillers, follow strict medical protocols, and provide clear guidance on treatment planning and pricing. You pay only for what you need. Dermal filler correction and reversal when needed.
We also provide dermal filler correction and reversal for select patients with prior filler.
What Dermal Fillers Can Treat
Dermal fillers can be used to address a wide range of aesthetic concerns while maintaining natural expression.
Lip Enhancement
Lip enhancement is one of the most requested dermal filler treatments. Whether you’re looking to restore volume lost over time or subtly refine shape and definition, treatment planning focuses on balance, proportion, and natural movement.
At our Sandy Springs practice, lip fillers are performed exclusively by Dr. Timani using conservative, anatomy-based techniques designed to:
• Restore natural lip volume
• Define the lip border
• Improve symmetry
• Maintain soft, natural expression
Cheek & Midface Volume
As facial volume changes over time, the midface may appear flatter or less supported. Strategic filler placement can restore structure, improve contour, and soften shadowing — while maintaining natural expression.
• Restore structural support
• Improve contour
• Reduce midface hollowing
Jawline & Chin Definition
The lower face plays a key role in overall facial balance. In appropriate candidates, dermal fillers may enhance chin projection or refine jawline contours to support proportion and profile harmony.
• Improve lower-face balance
• Enhance profile definition
• Refine contour conservatively
For patients seeking lifting without added volume, PDO thread lift treatments may be discussed during consultation.
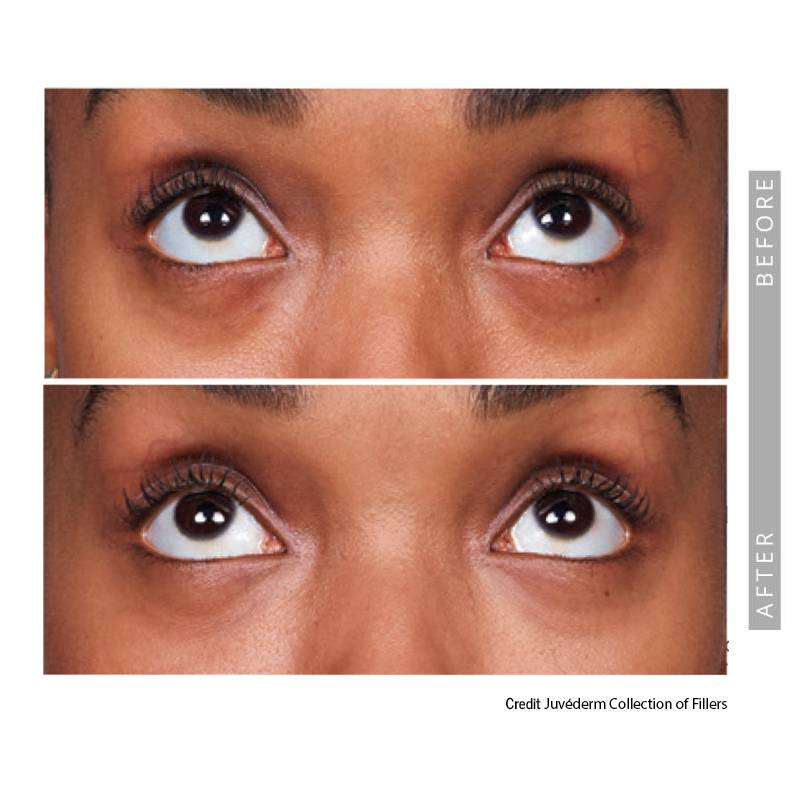
Under-Eye Hollows
Shadowing under the eyes can create a tired appearance. In carefully selected patients, filler may reduce hollowing and improve light reflection in this delicate area.
• Reduce tear trough shadowing
• Improve overall under-eye smoothness
Skin Hydration with SKINVIVE™
SKINVIVE™ is a hyaluronic acid micro-droplet treatment designed to improve skin smoothness and hydration rather than add volume.
• Improve skin texture
• Enhance hydration
• Subtle, refreshed appearance
Targeted Filler Treatments Beyond the Face (Selected Cases)
Some dermal fillers can also be used in select areas beyond the face, depending on your anatomy, skin quality, and goals. When appropriate, treatment may support subtle improvement in skin texture or volume-related concerns.
Areas that may be considered include:
• Décolletage lines
• Hands
• Certain types of body creasing or texture concerns (case-dependent)
Because product choice, depth, and technique vary widely, we’ll discuss what’s appropriate during your consultation — and whether another treatment option would be a better fit.
Book Your Dermal Filler Consultation
If you’re considering dermal fillers and want a conservative, physician-led plan, we’re here to help. Call us at (470) 769-9400 to schedule your consultation at Springs Dermatology MD in Sandy Springs, serving patients across Metro Atlanta.
Prefer North Atlanta? Visit our second office, Johns Creek Dermatology.
Dermal Fillers Frequently Asked Questions
What are dermal fillers?
Dermal fillers are FDA-approved injectable treatments designed to restore volume, enhance contours, and soften lines. Most fillers are composed of hyaluronic acid, a substance naturally found in the skin that supports hydration and structure.
Yes. Fillers are often combined with wrinkle relaxers suhc as Botox, PDO thread lifts, microneedling, or chemical peels to support balanced, natural-looking results. Your plan is personalized based on your anatomy, goals, and skin needs.
What is hyaluronic acid (HA)?
Hyaluronic acid is a naturally occurring molecule that helps the skin retain moisture and maintain elasticity. HA-based fillers provide immediate volume and are reversible if needed, offering flexibility and safety in treatment planning.
Will I still look like myself?
That is always the goal. At our practice, we follow a conservative, anatomy-based approach designed to enhance facial balance — not change your natural features. Results should look refreshed, structured, and harmonious.
When will I see results?
Results are typically visible immediately, although mild swelling may temporarily obscure the final outcome. As swelling subsides, the refined result becomes clearer.
How long do results last?
Longevity varies depending on the product used, treatment area, and individual metabolism. Many patients experience results lasting 6–18 months. During consultation, we discuss realistic expectations based on your goals.
Can fillers be combined with other treatments?
Yes. Fillers may also be part of a broader rejuvenation plan that includes microneedling for collagen support, Botox, PDO thread lifts, or chemical peels as part of a comprehensive facial rejuvenation plan. Treatment combinations are selected based on anatomy and long-term strategy.
Are dermal fillers safe?
When performed by a trained, board-certified dermatologist with deep anatomical knowledge, dermal fillers have a strong safety profile. All treatments include discussion of potential risks, benefits, and alternatives prior to injection.
Indications, Important Safety Information and Prescribing Information
All images © 2020 Allergan
JUVÉDERM® Collection of Fillers Important Information
INDICATIONS
JUVÉDERM® VOLUMA™ XC injectable gel is indicated for deep (subcutaneous and/or supraperiosteal) injection for cheek augmentation to correct age-related volume deficit in the mid-face in adults over the age of 21.
JUVÉDERM® VOLLURE™ XC injectable gel is indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds) in adults over the age of 21.
JUVÉDERM® Ultra Plus XC and JUVÉDERM® Ultra XC injectable gels are indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds).
JUVÉDERM® VOLBELLA™ XC injectable gel is indicated for injection into the lips for lip augmentation and for correction of perioral rhytids in adults over the age of 21.
JUVÉDERM® Ultra XC injectable gel is indicated for injection into the lips and perioral area for lip augmentation in adults over the age of 21.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
These products should not be used in patients who have severe allergies, marked by a history of anaphylaxis or history or presence of multiple severe allergies, and should not be used in patients with a history of allergies to Gram-positive bacterial proteins or lidocaine contained in these products.
WARNINGS
- Do not inject into blood vessels. Introduction of these products into the vasculature may lead to embolization, occlusion of the vessels, ischemia, or infarction. Take extra care when injecting soft-tissue fillers; for example, inject the product slowly and apply the least amount of pressure necessary. Rare, but serious, adverse events associated with the intravascular injection of soft-tissue fillers in the face have been reported and include temporary or permanent vision impairment, blindness, cerebral ischemia or cerebral hemorrhage leading to stroke, skin necrosis, and damage to underlying facial structures. Immediately stop the injection if a patient exhibits any of the following symptoms: changes in vision, signs of a stroke, blanching of the skin, unusual pain during or shortly after the procedure. Patients should receive prompt medical attention and, possibly, evaluation by an appropriate healthcare professional specialist should an intravascular injection occur
- Product use at specific sites in which an active inflammatory process (skin eruptions such as cysts, pimples, rashes, or hives) or infection is present should be deferred until the underlying process has been controlled
PRECAUTIONS
- To minimize the risk of potential complications, these products should only be used by healthcare professionals who have appropriate training, experience, and knowledge of facial anatomy
- Healthcare professionals are encouraged to discuss the potential risks of soft-tissue injections with their patients prior to treatment and ensure that patients are aware of signs and symptoms of potential complications
- The safety and effectiveness for the treatment of anatomic regions other than the mid-face with JUVÉDERM® VOLUMA™ XC; facial wrinkles and folds with JUVÉDERM® Ultra XC, JUVÉDERM® Ultra Plus XC, and JUVÉDERM® VOLLURE™ XC; and the lips and perioral area with JUVÉDERM® Ultra XC and JUVÉDERM® VOLBELLA™ XC have not been established in controlled clinical studies
- As with all transcutaneous procedures, dermal filler implantation carries a risk of infection. Follow standard precautions associated with injectable materials
- The safety for use during pregnancy, in breastfeeding females, and in patients with known susceptibility to keloid formation, hypertrophic scarring, and pigmentation disorders has not been studied
- The safety for use of JUVÉDERM® VOLUMA™ XC in patients under 35 or over 65 years, JUVÉDERM® Ultra XC and JUVÉDERM® Ultra Plus XC in patients under 18 years, and JUVÉDERM® VOLLURE™ XC and JUVÉDERM® VOLBELLA™ XC in patients under 22 years has not been established
- Use with caution in patients on immunosuppressive therapy
- Patients who are using products that can prolong bleeding (such as aspirin, nonsteroidal anti-inflammatory drugs, and warfarin) may experience increased bruising or bleeding at treatment sites
- If laser treatment, chemical peel, or any other procedure based on active dermal response is considered after treatment, or if these products are administered before the skin has healed completely, there is a possible risk of an inflammatory reaction at the treatment site
- Patients who experience skin injury near the site of implantation may be at a higher risk for adverse events
- The safety of JUVÉDERM® VOLUMA™ XC injectable gel for use in patients with very thin skin in the mid-face and Fitzpatrick Skin Types V and VI has not been established
- Patients may experience late onset nodules with use of dermal fillers including JUVÉDERM® VOLUMA™ XC
- Patients may experience late onset adverse events with use of dermal fillers
ADVERSE EVENTS
The most commonly reported side effects for JUVÉDERM® injectable gels were injection-site redness, swelling, pain, tenderness, firmness, lumps/bumps, bruising, discoloration, and itching. For JUVÉDERM® VOLBELLA™ XC, dryness was also reported. For JUVÉDERM® VOLUMA™ XC, side effects were predominantly moderate in severity, with duration of 2 to 4 weeks; for JUVÉDERM® Ultra XC , JUVÉDERM® Ultra Plus XC, or JUVÉDERM® VOLLURE™ XC, they were mostly mild or moderate in severity, with duration of 14 days or less; and for JUVÉDERM® VOLBELLA™ XC, they were predominantly mild or moderate, with duration of 30 days or less.
To report an adverse reaction with any product in the JUVÉDERM® Collection, please call Allergan at 1-800-433-8871. Please visit JuvedermDFU.com for more information.
Products in the JUVÉDERM® Collection are available by prescription only.
Peggy Wood - Google Review
Dr Timani is an artist. Providing state of the art treatments that are natural looking. Never overdone. She is a perfectionist sand takes her time to make each treatment perfect.
PATIENT REVIEWS