Raj Joshi - Go to review
Dr. Timani is professionally very knowledgeable and kind. Her team is also dedicated to customer service. The whole process from front desk to check out is very smooth.PATIENT REVIEWS


Indications, Important Safety Information and Prescribing Information
All images © 2020 Allergan
KYBELLA®
KYBELLA® (deoxycholic
acid) injection 10 mg/mL Important Information
INDICATION
KYBELLA® (deoxycholic
acid) injection is indicated for improvement in the appearance of moderate to severe
convexity or fullness associated with submental fat in adults.
The
safe and effective use of KYBELLA® for the treatment of
subcutaneous fat outside the submental region has not been established and is
not recommended.
IMPORTANT
SAFETY INFORMATION
CONTRAINDICATIONS
KYBELLA® is
contraindicated in the presence of infection at the injection sites.
WARNINGS
AND PRECAUTIONS
Marginal
Mandibular Nerve Injury
Cases
of marginal mandibular nerve injury, manifested as an asymmetric smile or
facial muscle weakness, were reported in 4% of subjects in the clinical trials;
all cases resolved spontaneously (range 1-298 days, median 44 days). KYBELLA®
should not be injected into or in close proximity to the marginal mandibular
branch of the facial nerve.
Dysphagia
Dysphagia
occurred in 2% of subjects in the clinical trials in the setting of
administration-site reactions, eg, pain, swelling, and induration of the
submental area; all cases of dysphagia resolved spontaneously (range 1-81 days,
median 3 days). Avoid use of KYBELLA® in patients with current
or prior history of dysphagia as treatment may exacerbate the condition.
Injection-Site
Hematoma/Bruising
In
clinical trials, 72% of subjects treated with KYBELLA® experienced
hematoma/bruising. KYBELLA® should be used with caution in
patients with bleeding abnormalities or who are currently being treated with
antiplatelet or anticoagulant therapy as excessive bleeding or bruising in the
treatment area may occur.
Risk
of Injecting into or in Proximity to Vulnerable Anatomic Structures
To
avoid the potential of tissue damage, KYBELLA® should not be
injected into or in close proximity (1 cm-1.5 cm) to salivary glands, lymph
nodes, and muscles. Care should be taken to avoid inadvertent injection
directly into an artery or a vein as it can result in vascular injury
Injection
Site Alopecia
Cases
of injection site alopecia have been reported with administration of KYBELLA®.
Onset and duration may vary among individuals and may persist. Consider
withholding subsequent treatments until resolution.
Injection
Site Ulceration and Necrosis
Injections
that are too superficial into the dermis may result in skin ulceration and
necrosis. Cases of injection site ulceration and necrosis have been reported
with administration of KYBELLA®. Do not administer KYBELLA® into
affected area until complete resolution.
ADVERSE
REACTIONS
The
most commonly reported adverse reactions in the pivotal clinical trials were:
injection site edema/swelling, hematoma/bruising, pain, numbness, erythema, and
induration.
Please
see KYBELLA® full Prescribing Information.
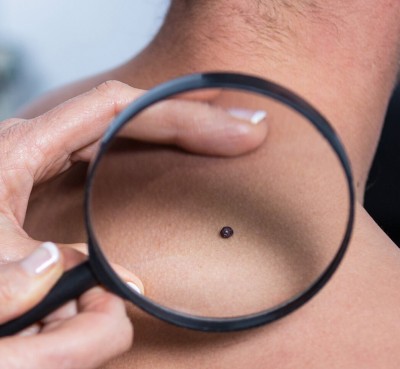
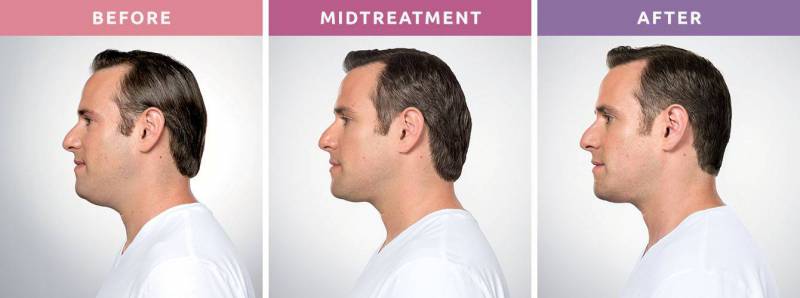
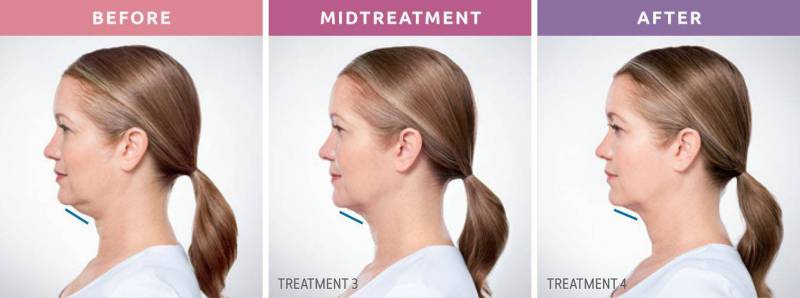
Say goodbye to that double chin - for good!
A Non-Surgical Double Chin Removal
Struggling with stubborn fat under your chin that won’t go away, no matter how much you diet or exercise? A double chin can affect your confidence and profile, but there’s good news: Kybella® offers a non-surgical, FDA-approved solution to permanently eliminate those fat cells. It’s the first and only injectable treatment designed to reduce submental fullness, helping you achieve a slimmer, more defined jawline without surgery or downtime.
Why Choose Springs Dermatology MD for Kybella®
At Springs Dermatology MD, we deliver precise, patient-focused Kybella® treatments in Sandy Springs and Metro Atlanta, ensuring safe, effective, and natural-looking results tailored to your needs. Here's what sets us apart!
Expert Dermatologists
Our board-certified team has extensive experience in delivering safe and effective Kybella® treatments.
Customized Plans
Every chin profile is unique, so we create personalized treatment plans to meet your aesthetic goals.
Combined Treatments
Enhance your results by pairing Kybella® with NovaThreads for a non-surgical chin or neck lift. Patient-
Centered Approach
We carefully explain the process, risks, and benefits, ensuring you feel confident every step of the way.
Proven Results
Using FDA-approved Kybella®, we provide a solution that’s scientifically backed for permanent fat reduction.
Ready to redefine your profile? Schedule your Kybella® consultation today to eliminate fat cells under your chin and reveal a more defined, youthful profile.
How Kybella® Works
Kybella® uses synthetic deoxycholic acid, a naturally occurring molecule in your body that breaks down dietary fat. When injected into the fat beneath the chin, Kybella®:
Eliminates Fat Cells
These cells can no longer store or accumulate fat once removed.
Improves Your Profile
Gradually reveals a slimmer, more contoured appearance.
Offers Permanent Results
Once the fat cells are gone, they’re gone for good.
What to Expect From Your Treatment
Visible Results
Start seeing changes within 2–4 treatments, with full results typically visible in 3 months.
Minimal Downtime
Swelling, bruising, or numbness may occur for a few days but usually resolves quickly.
Quick Sessions
Each session lasts about 15–20 minutes, fitting seamlessly into your busy schedule.
Benefits of Kybella
Non-Surgical Solution
No need for invasive procedures or downtime.
FDA-Approved
Safe and effective for adults with moderate to severe submental fat. Enjoy a sharper, more defined jawline without surgery.
Permanent Results
Achieve your desired profile with lasting results after completing your treatment plan.
Frequently Asked Questions
What is submental fullness?
Submental fullness is a gathering of excess fat beneath the chin, commonly known as “double chin.” It can be something you’ve had your whole life due to genetics or start to appear as you get older or if you gain weight.
What is Kybella?
The active ingredient in KYBELLA is synthetic deoxycholic acid. Deoxycholic acid is a naturally occurring molecule in the body that aids in the breakdown and absorption of dietary fat. When injected into the fat beneath the chin, KYBELLA destroys fat cells and once destroyed, these cells can no longer store or accumulate fat. This results in a noticeable reduction in fullness under the chin, revealing an improved chin profile.
When will I see the results?
Visible results in 2-4 treatments. The results of KYBELLA injections happen gradually. You will start noticing changes in your appearance within 4 to 6 weeks. However, it typically takes about 3 months before you can see the full results.
How long do the results last?
The results are permanent. Once dissolved, these cells can no longer store or accumulate fat, so further treatment is not expected once you reach your desired aesthetic goal.
How many treatments will I need?
Since everyone’s chin profile is different, the number of treatments varies from patient to patient.
Does it hurt?
Each patient will have their own pain threshold. Patients have reported that KYBELLA® injections are no more than a mild burning sensation for around 15 minutes. We apply ice packs for this time to maximize our patients' comfort.
What is the downtime?
After KYBELLA treatment, you will likely have some swelling, bruising, or numbness under your chin that will subside within 2 to 3 days. Cold compresses may be used to reduce the swelling.
Is Kybella safe?
KYBELLA is a prescription medicine and is the only FDA-approved injectable treatment used in adults to improve the appearance and profile of moderate to severe fat below the chin (submental fat), also called “double chin.”
For more information: https://www.mykybella.com/
Indications, Important Safety Information and Prescribing Information
All images © 2020 Allergan
KYBELLA®
KYBELLA® (deoxycholic
acid) injection 10 mg/mL Important Information
INDICATION
KYBELLA® (deoxycholic
acid) injection is indicated for improvement in the appearance of moderate to severe
convexity or fullness associated with submental fat in adults.
The
safe and effective use of KYBELLA® for the treatment of
subcutaneous fat outside the submental region has not been established and is
not recommended.
IMPORTANT
SAFETY INFORMATION
CONTRAINDICATIONS
KYBELLA® is
contraindicated in the presence of infection at the injection sites.
WARNINGS
AND PRECAUTIONS
Marginal
Mandibular Nerve Injury
Cases
of marginal mandibular nerve injury, manifested as an asymmetric smile or
facial muscle weakness, were reported in 4% of subjects in the clinical trials;
all cases resolved spontaneously (range 1-298 days, median 44 days). KYBELLA®
should not be injected into or in close proximity to the marginal mandibular
branch of the facial nerve.
Dysphagia
Dysphagia
occurred in 2% of subjects in the clinical trials in the setting of
administration-site reactions, eg, pain, swelling, and induration of the
submental area; all cases of dysphagia resolved spontaneously (range 1-81 days,
median 3 days). Avoid use of KYBELLA® in patients with current
or prior history of dysphagia as treatment may exacerbate the condition.
Injection-Site
Hematoma/Bruising
In
clinical trials, 72% of subjects treated with KYBELLA® experienced
hematoma/bruising. KYBELLA® should be used with caution in
patients with bleeding abnormalities or who are currently being treated with
antiplatelet or anticoagulant therapy as excessive bleeding or bruising in the
treatment area may occur.
Risk
of Injecting into or in Proximity to Vulnerable Anatomic Structures
To
avoid the potential of tissue damage, KYBELLA® should not be
injected into or in close proximity (1 cm-1.5 cm) to salivary glands, lymph
nodes, and muscles. Care should be taken to avoid inadvertent injection
directly into an artery or a vein as it can result in vascular injury
Injection
Site Alopecia
Cases
of injection site alopecia have been reported with administration of KYBELLA®.
Onset and duration may vary among individuals and may persist. Consider
withholding subsequent treatments until resolution.
Injection
Site Ulceration and Necrosis
Injections
that are too superficial into the dermis may result in skin ulceration and
necrosis. Cases of injection site ulceration and necrosis have been reported
with administration of KYBELLA®. Do not administer KYBELLA® into
affected area until complete resolution.
ADVERSE
REACTIONS
The
most commonly reported adverse reactions in the pivotal clinical trials were:
injection site edema/swelling, hematoma/bruising, pain, numbness, erythema, and
induration.
Please
see KYBELLA® full Prescribing Information.
Raj Joshi - Go to review
Dr. Timani is professionally very knowledgeable and kind. Her team is also dedicated to customer service. The whole process from front desk to check out is very smooth.PATIENT REVIEWS